Microsoft engineer one of the first to test coronavirus vaccine for COVID-19
2 min. read
Published on
Read our disclosure page to find out how can you help MSPoweruser sustain the editorial team Read more
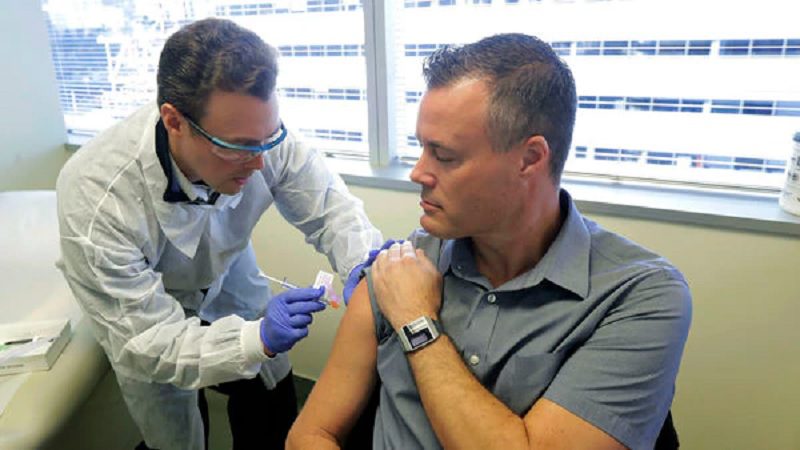
Clinical trials of a coronavirus vaccine being developed for the Coronavirus pandemic has started yesterday in the USA, and a Microsoft network engineer has been one of the first to receive the experimental product.
Neal Browning, 46, of Bothell, Washington, is a Microsoft network engineer with young daughters.
“Every parent wants their children to look up to them,” he said. But he’s told them not to brag to their friends. “It’s other people, too. It’s not just Dad out there.”
Browning is part of a group of four healthy volunteers who are testing a new RNA COVID-19 vaccine developed by NIH and Massachusetts-based biotechnology company Moderna Inc. and being tested by Kaiser Permanente Washington Research Institute in Seattle. Moderna developed the vaccine at an unprecedented speed only 65 days after the virus was sequenced in China.
“We’re team coronavirus now,” Kaiser Permanente study leader Dr. Lisa Jackson said on the eve of the experiment. “Everyone wants to do what they can in this emergency.”
If all goes well the next step is to give 45 volunteers two doses, a month apart, at various doses, to check their response.
Despite the rapid process the team only expects to have a vaccine widely available in 12 to 18 months.
Because vaccines are given to millions of healthy people, it takes time to test them in large enough numbers to spot an uncommon side effect, cautioned Dr. Nelson Michael of the Walter Reed Army Institute of Research, which is developing a different coronavirus vaccine candidate.
“The science can go very quickly but, first, do no harm, right?” he told reporters last week.
Notable other researchers have suggested a vaccine could be available much sooner, with a team from the University of Queensland suggesting they could have a vaccine on the market by the end of the year.




User forum
0 messages